To understand the chemistry of cheese, we must first understand the chemistry of milk because cheese is made from milk. Milk is “an emulsion of milk fat globules in an aqueous (watery) environment.”
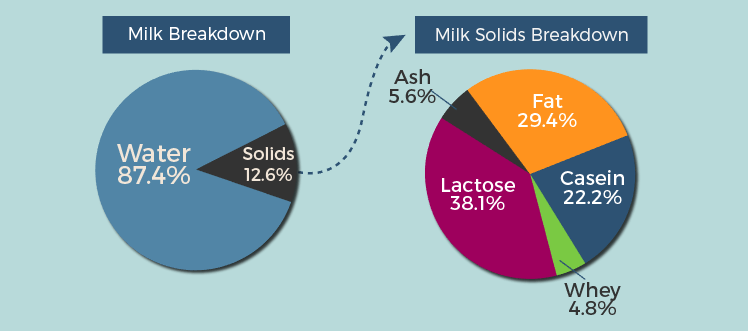
Milk is made mostly of water (87.4%) and the rest is solids (12.6%). Of these solids, the major components are lactose (milk sugar), fat, and protein (casein and whey). To make cheese, one must remove the water from milk, concentrating the fat and protein.
Lactose is a disaccharide which means it is made of two monosaccharides (glucose and galactose). In the cheesemaking process, bacterial cultures are added which consume lactose and produce lactic acid. With aged cheeses, all lactose is converted to lactic acid. This explains why lactose intolerant people can eat aged cheeses without much problem.
Fat exists in globular structures; these globules have multiple layers and membranes. When cheese is made, the fat is concentrated and the water is removed from the milk. This process allows us to see the yellowish hue in cheese. In milk, the water is what gives it a white color. The yellow color comes from betacarotene that cows get from eating grass.
Milk contains two types of proteins (casein and whey). The main type of casein proteins are alpha s-1, alpha s-2, beta, and kappa. The whey is the watery remainder left out of the cheese. Kappa casein are important in the cheese making process because they must stick together to form curd from milk. Casein proteins are negatively charged at neutral pH and neutral at low pH and so they stick together to form curds. Rennet is used to get milk to form curd; it is composed of enzymes found in calf stomach.